
Me and King (Talebreed horse)
Hey! I just started O-level in training horse! My dean introduced this to me and why not? Even if I am not into horses (they are super big and I'm just a 'ketot girl') but I took this opportunity to learn and suddenly fell in love with it. The class starts during weekend (so can u imagine how busy i am?) and damnn tired! but I enjoyed the my first day of class and hopefully can keep it up until I am good enough to ride. Maybe having an equine license can make u proud instead of a car license ..right?
Post today is about disease of horses, particularly in GIT. Actually this is part of my assignment during clinical biology class. Haha
************************************************
Clostridium Perfingens
Clostridium perfringens is an anaerobic, Gram-positive, sporeforming rod (anaerobic means unable to grow in the presence of free oxygen). It is widely distributed in the environment and frequently occurs in the intestines of humans and many domestic and feral animals. Spores of the organism persist in soil, sediments, and areas subject to human or animal fecal pollution.
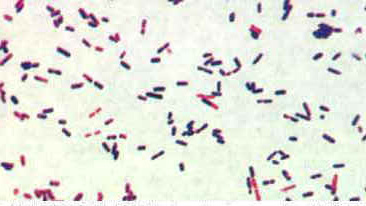
C. Perfingens
Infections due to C. Perfingens causes tissue necrosis, bacteremia, emphysematous cholecystitis, and gas gangrene, which is also known as clostridial myonecrosis. The toxin involved in gas gangrene is known as α-toxin, which inserts into the plasma membrane of cells, producing gaps in the membrane that disrupt normal cellular function. C. perfringens can participate in polymicrobial anaerobic infections. Clostridium perfringens is commonly encountered in infections as a component of the normal flora. In this case, its role in disease is minor.
C. perfringens is an anaerobic bacterium, who acquires energy by performing anaerobic respiration using Nitrate as its electron acceptor. There is an increase in growth when this bacterium is grown in the presence of Nitrate, because this inorganic acceptor allows more metabolites molecules to undergo substrate-level phosphorylation reactions, leading to an increase yield in energy production. C. perfringens can also undergo anaerobic fermentation to produce gases, such as carbon dioxide, that may increase its survival by creating a preferred anaerobic habitat in host tissues. C. perfringens also has all the enzymes necessary to carry out glycolysis and glycogen metabolism. However, C. perfringens does not have a complete set of genes necessary for amino acid biosynthesis; in fact, only 45 enzyme-encoding genes were discovered. Therefore, C. perfringens cannot survive on media that are lacking an essential amino acid supply.
Pathology
C. perfringens is categorized into five serotypes—A, B, C, D, and E—depending on the types of extracellular toxins (alpha-, beta, epsilon-, and iota-toxins) they make and their forms of tropisms. C. perfringens is a pathogen whose primary targets are human and animals. The bacterium can be found in many different habitats, such as the normal flora of human gastrointestinal (GI) tract, and environment, such as sewage and soil. Several common diseases associated with C. perfringens are food-poisoning, gas gangrene, and many veterinary diseases. C. perfringens enterotoxin (CPE) is the main virulent factor that initiates many critical GI diseases. When food contaminated with C. perfringens is consumed, CPE begins its membrane action in a unique four-step mechanism.
1. Binding CPE to the target receptor on plasma membrane protein or claudin proteins, which leads to the formation of a small complex.
2. The complex then undergoes physical change when it binds to other membrane proteins and forms a larger complex in the membranes, which results in the disruption of the membrane’s permeability. This usually leads to cell death, because the osmotic equilibrium is not maintained due to the breakdown of the membrane’s permeability
3. CPE is capable of forming a larger complex in the membrane and its toxic level is greatly enhanced when the first 45 N-terminal amino acids are eliminated.
4. On the other hand, eliminating amino acids outside of residue 45 prohibits CPE from forming large complex, and thus loses its toxicity.
A range of veterinary enterotoxaemias are caused by toxins absorbed from the intestinal bacteria. These include; necrotic enteritis in poultry and fowl (type A), an enterotoxaemic jaundice in lambs (type A), lamb dysentery (type B), an enterotoxaemia in neonatal calves and foals (type B), a hemorrhagic enterotoxaemia in piglets, calves and foals (type C), Struck in adult sheep (type C), Pulpy kidney disease in sheep, goats and calves (type D) and a rare enterotoxaemia in calves and lambs (type E).
Clostridia-associated enterocolitis in horse

Clostridium difficile and C perfringens have been implicated in this acute, sporadic disease of horses characterized by diarrhea and colic. Because of uncertainty about the etiology, the condition has also been referred to as idiopathic colitis, but there is now good evidence that these organisms are responsible for enterocolitis in horses.
C difficile is found only infrequently and C perfringens is found in low concentrations in the feces of normal horses. Both organisms may be present in soil or the environment and be ingested by horses. The factors that trigger disease are not well known, but it is presumed that some alteration in the normal flora permits excessive multiplication of the bacteria, which produce toxins capable of causing intestinal damage and systemic effects.
Predisposing factors that have been suggested include change in diet and antibiotic therapy. Other host factors that may determine whether disease develops include age, immunity, and presence or absence of intestinal receptors for the clostridial toxins. C difficile produces protein toxin A or B or both in the intestine. Toxin A is an enterotoxin that causes hypersecretion of fluid into the intestinal lumen and also causes tissue damage. Toxin B is a potent cytotoxin that induces inflammation and necrosis.
Recent antibiotic therapy is a common feature of the history of horses with C difficile -induced diarrhea. Certain antibiotics, notably erythromycin, β-lactam antibiotics, and trimethoprim/sulfonamide, are more likely than others to be associated with C difficilecolitis. Mares with foals that are being treated with erythromycin appear to be at high risk.
Elimination of roughage from the diet prior to surgery is also reported to predispose to C difficile colitis.C perfringens type A is believed to cause diarrhea by elaboration of an enterotoxin (CPE), which is released during sporulation and stimulates intestinal epithelial cells to secrete excess fluid into the lumen. A novel necrotizing toxin, called β2, produced by some strains of C perfringens , has recently been strongly associated with colitis in horses.
The characteristic lesion is a necrotizing enterocolitis. There is severe loss of colonic and cecal mucosal epithelial cells, hemorrhagic colitis and typhlitis, and thrombosis in capillaries of the intestinal mucosa.

Clinical signs include sudden death, diarrhea with or without blood, colic, reduced feed intake, and lethargy. These clinical signs are also consistent with other causes of enterocolitis. Foals affected at <3 days old with C perfringens -associated enterocolitis often have bloody diarrhea and colic. C perfringens type C infection in neonatal foals has consistently been associated with severe GI disease. Several foals on a particular farm may be affected, but the disease is typically sporadic. The role of C perfringens type A in enterocolitis in neonatal foals is less clear; it has been reported that >90% of foals at 3 days of age shed this organism in their feces and that C perfringens type A is likely one of the first bacteria to colonize the intestinal tract of newborn foals, irrespective of hygiene protocols. C difficile has been associated with enterocolitis in newborn foals as well as in adult horses. It has been identified as a nosocomial infection in humans, and this may also be seen in horses.

Foals and adult horses may be affected. Typically there are signs of abdominal pain and diarrhea with or without blood. There may be abdominal distention, especially in cases of C difficile -induced diarrhea. Dehydration, toxemia, and shock may develop, and the mortality rate is variable. One or several animals on a farm may be affected.
Diagnosis
Clinical features of the disease are similar to those of acute salmonellosis, Potomac horse fever, or monocytic ehrlichiosis. The identification of C perfringens as the cause of diarrhea in horses depends on demonstration of the presence of enterotoxin or the gene for CPE in the feces or intestinal fluid and the absence of other likely etiologic agents. Most C perfringens found in the intestine of horses lack the gene for CPE. Demonstration of large numbers of bacterial spores or a high concentration of C perfringens in the feces are also aids to a diagnosis.
Control
Metronidazole and chloramphenicol for oral administration to high-risk horses is recommended. The sources of C difficile spores may be attacked by surface disinfection with a sporicidal disinfectant, and the spread may be reduced by hand washing and by isolation of infectious horses and foals. Supportive care by using IV polyionic fluids, supplemental electrolytes, antiinflammatories, and broad spectrum of antibiotics can be used if the horse is leukopenic and at risk of bacterial translocation across the compromised GI tract.
*Recent finding: The yeast of Saccharomyces boulardii has been shown to be protective in clostridia diarrhea by degrading C difficile toxins A and B.
Sources: Clostridia.net, C.Perfringens microbewiki.kenyon.edu, The Merck veterinary manual




















2 comments:
Hi Aina!
do you know if this kind of diorrhea is associated to strong sour smell, yellowish feces, fast loose of weigth, lethargy and highly percentage of transmission?
I am asking due to the death of a beloved 2-months-old foal. It was an orphan, female and was the only one who died among her 7 companions (5 affected by the diorrhea, 2 of them severely).
thanks a lot.
Nice effort to write about this. here is my veterinary medicine blog
Visit Veterinary Medicine!
Post a Comment